At MME Nordic we help our customers meet the requirements related to quality, documentation and validation, which the medico Industry is subject to

Medical equipment must meet essential safety and performance regulations to meet all safety requirements from authorities, patients and users. If this trust is violated, it can lead to a sudden drop in sales of the product concerned.
At MME Nordic we help our customers meet the requirements for quality, documentation and validation, which the medico Industry is subject to - in a structured and manageable way.
We perform quality management in all assignments within the certified ISO 9001:2015 management system. We construct machines that meet the international guidelines within GxP and GAMP 5.
At MME Nordic we relate to the following guidelines within GxP and GAMP 5:
At MME Nordic we help our customers meet the requirements related to quality, documentation and validation, which the medico Industry is subject to
We know that the overall guidelines within GxP and GAMP 5 form a foundation for handling procedures in many ways. Therefore, we ensure that all requirements are met, and we make sure that any uncertainty is taken care of.
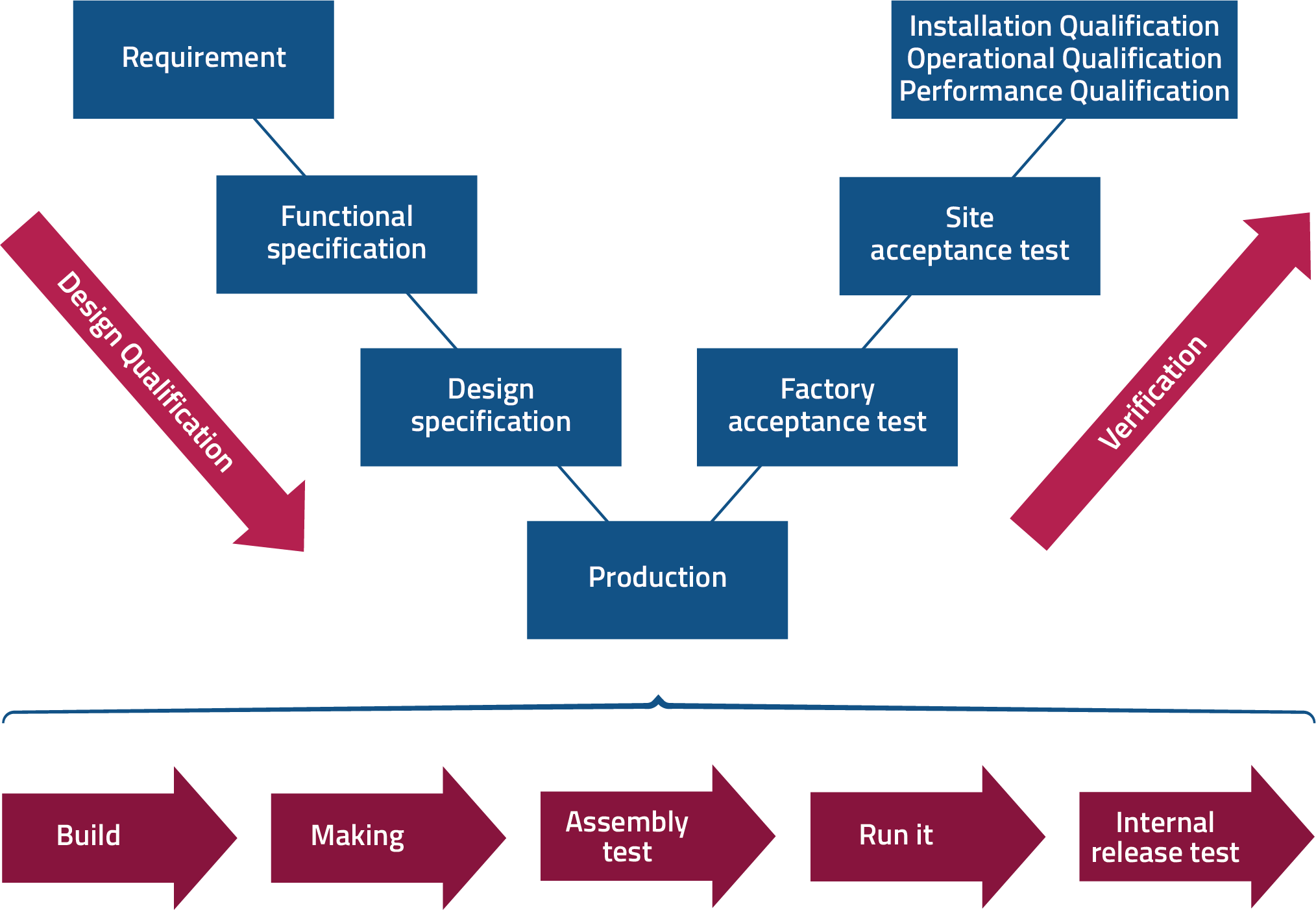
At MME Nordic we have extensive knowledge within procedures and processes in the V-model, which we use a foundation of our work. Therefore, we offer a very structured and detailed documentation of the whole process from user requirement specifications to design and software specifications, FAT and SAT tests as well as the following validation within the three phases, IQ, OQ and PQ, if necessary. This ensures that the design, functionality, and requirements are thoroughly tested and documented before being put into use.
All tests are carried out according to agreed terms, and as customer you will always be invited to witness the tests when they are being carried out. Test plans, protocols and reports are handled in accordance with agreed terms mentioned before.
Our structured approach to documentation gives you the opportunity to follow the ongoing tests, we conduct on your machine.
Together with documentation and construction of the machine, we also handle the format of user guides and maintenance requirements as well as CE Marking.

As a manufacturer it is important to know the possible risks on the machine as well as the final product. We already take these risks into account in the URS phase where we compose a thorough FMEA analysis.
We ensure clear traceability in both the requirements as well as the implementation. We create a separate and oversee able document. This makes it easy to identify the requirements.